
Scientists have developed a model of the early stages of Alzheimer’s disease in rhesus macaques to better test new experiments.

Researchers at the California National Primacy Research Center (CNPRC) at the University of California, Davis, USA, have developed a model of the early stages of Alzheimer’s disease in rhesus macaques. The new macaque model could better test novel treatments.
According to the researchers, understanding what happens during the progression of Alzheimer’s disease may be important in preventing or reversing the symptoms of the condition. However, it is difficult to study therapeutic strategies without an effective animal model that closely resembles the human condition. Much research has focused on transgenic mice expressing a human version of amyloid or tau proteins, but these studies have found it difficult to translate into new therapies.
The team says that humans and monkeys have two types of tau protein in the brains, but rodents only have one.
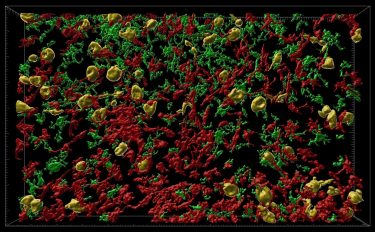
Inflammatory cells (red and green) flow into a part of the brain where tau neurons are under the influence of tau protein (orange). This model in rhesus macaque monkeys shows similarities to the development of Alzheimer’s disease in humans and could open up ways to try new treatments [credit: Danielle Beckman, UC Davis/CNPRC].
“We think the macaque is a better model, because it expresses the same versions of tau in the brain as humans,” said Danielle Beckman, a CNPRC postgraduate researcher and first author of the paper.
Mice also lack specific areas of the neocortex such as the prefrontal cortex, an area of the human brain that is highly vulnerable to Alzheimer’s disease. Pre-horizontal cortex is present in rhesus macaques and is extremely important for mental functions both in humans and in monkeys. Therefore, there is an urgent need for new and improved animal models for Alzheimer’s disease that can stand between mouse models and human clinical trials.
The researchers created versions of the human tau gene with mutations that cause misalignment, wrapped in viral material. These vectors have been introduced into the macas rhesus, in a brain area called the entorhinal cortex, which is highly vulnerable in Alzheimer’s disease.
Within three months, they could see that undigested tau proteins had spread to other parts of the animal’s brain. They found a mismatch of both introduced human tau mutant proteins and of the tau proteins themselves.
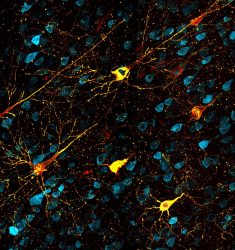
Neurons affected by complex tau proteins. Healthy neurons are blue, tau proteins are defective yellow and protein tangles are red. This model in rhesus macaque monkeys shows similarities to the development of Alzheimer’s disease in humans and could open up ways to try new treatments [credit: Danielle Beckman, UC Davis/CNPRC].
“The pattern of transmission vaguely showed that tau-based pathology followed the precise connections of the entorhinal cortex and that the proliferation of tau pathology could pass from one area to the next through synaptic connections,” said Dr John Morrison, lead researcher. “This ability to spread through brain circuits leads to damage to cortical areas that are dependent on higher level experience far from the entorhinal cortex.”
“We think this represents a lower level, but before cell death occurs far and wide,” Beckman said.
The next plan researchers will test whether behavioral changes similar to human Alzheimer’s disease develop in the rhesus macaque model. If so, it could be used to test for prevention of inflammation or inflammation.
The study was published in Alzheimer’s & Dementia: Journal of the Alzheimer’s Society.