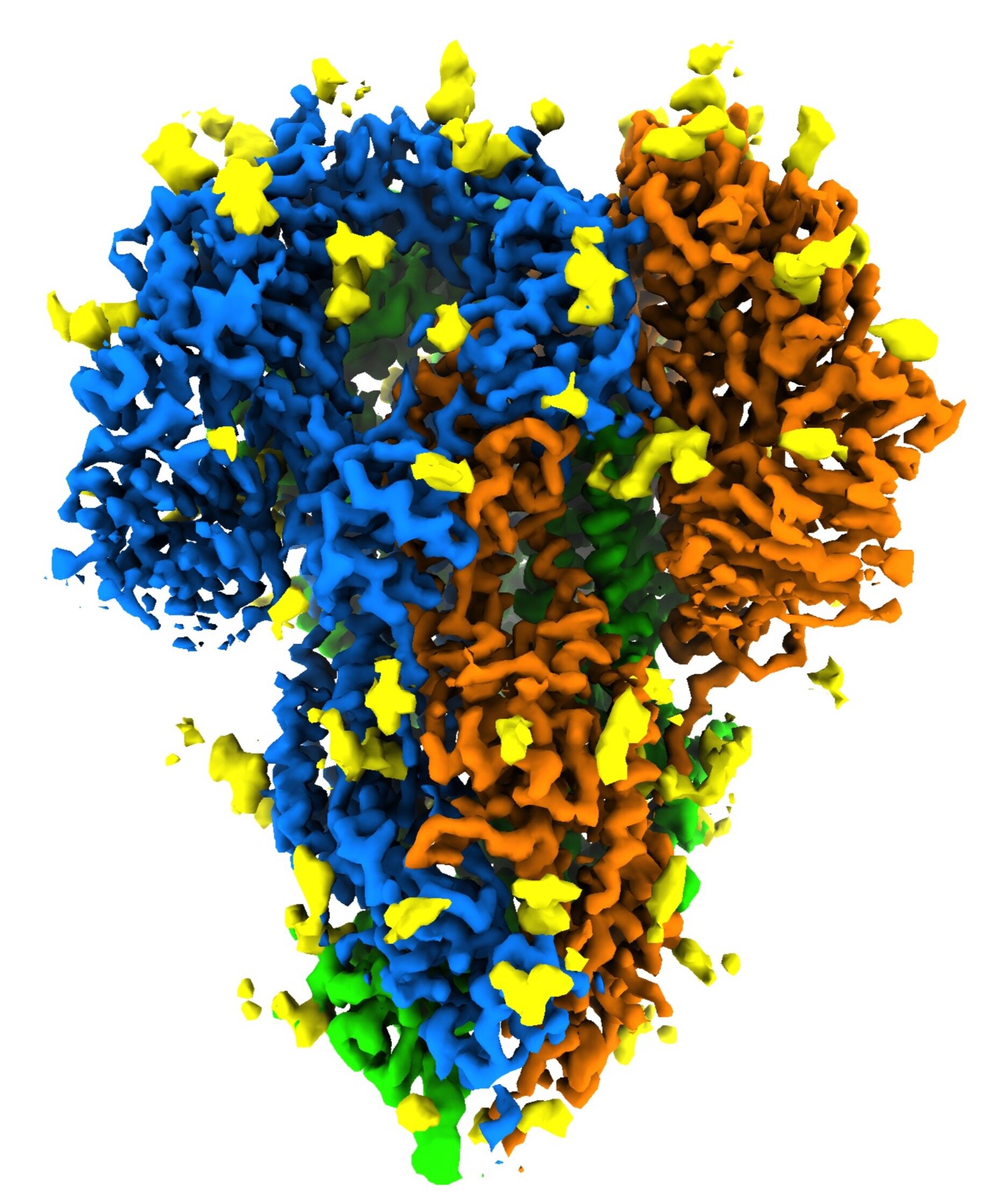

This image shows the detailed “spike” structure of a coronavirus that causes cold symptoms – a milder relative strain of the virus that causes COVID-19. Spikes attach to receptors on their victim’s cells to initiate infections. They contain three protein molecules, shown in blue, green and orange, full of sugar (yellow) molecules that play an important role in the life cycle of the virus and have the ability to bypass the immune system. Scientists at SLAC National Acceleration Laboratory and Stanford University created the image with a new method that shows spikes in their natural state, producing faster and more accurate images of the infectious device. Credit: K. Zhang et al., Quarterly Reviews of Biophysics Discovery, 2020
Coronaviruses as the cause of COVID-19 are full of protein “spikes” that bind to receptors on their sufferer’s cells – the first step in infection. Now scientists have made the first detailed images of these spikes in their natural state, while still being attached to the virus and without the use of chemicals that could shape them. turn.
They say their method, which combines a cryogenic electron microscope (cryo-EM) and a computational system, should produce faster and more accurate images of the infectious mechanism in different strains of coronavirus , an essential step in the design of drugs and therapeutic vaccines.
“The advantage of doing it this way is that when you clean a spike protein and study it on its own, you lose an important biological context: How it looks in part complete viral? It could have a different structure there, “said Wah Chiu, a professor at DOE SLAC ‘s national acceleration laboratory at DOE and Stanford University and senior author of the study. They described their findings in Quarterly Reviews Discover Biophysics.
Seven strains of coronaviruses are known to affect humans. Four cause relatively mild illnesses; the other three – including SARS-CoV-2, the virus that causes COVID-19 – can be deadly, said co-author Jing Jin, an expert in molecular biology of viruses at the Institute Vitalant Research in San Francisco. Scientists at Vitalant hunt for viruses in human and animal blood and stool samples, screen blood samples at times such as the common pandemic, and study interactions between viruses and their hosts .

This rotating image shows the detailed “spike” structure of a coronavirus that causes cold symptoms – a milder relative strain of the virus that causes COVID-19. Spikes attach to receptors on their victim’s cells to initiate infections. They contain three protein molecules, shown in blue, green and orange, full of sugar (yellow) molecules that play an important role in the life cycle of the virus and have the ability to bypass the immune system. Scientists at the SLAC National Accelerator Laboratory and Stanford University created the image with a new method that shows spikes in their natural state, producing faster and more accurate images of the infectious device. Credit: K. Zhang et al., Quarterly Reviews of Biophysics Discovery, 2020
The virus that causes COVID-19 is so robust that only a handful of cryo-EM laboratories in the world can diagnose it with a high enough level of biosecurity controls, Jin said. So for this study, the research team looked at a much milder coronavirus strain called NL63, which causes common cold symptoms and is responsible for about 10% of human respiratory disease each year. It is thought to bind to the same receptors on human cell surfaces as the COVID-19 virus.
Instead of chemically removing and purifying NL63 spike proteins, the researchers spray whole, intact viruses into a glassy state that preserves the natural alignment of their components. They then made thousands of detailed images of random viruses using cryo-EM instruments at Stanford-SLAC Cryo-EM Resources, digitally extracted the fragments containing spike proteins, and combined them. to get high-resolution images.
“The structure we saw had the same structure as it has on the surface of the virus, free of chemicals,” Jin said. “This has not been done before.”
The team also identified places where sugar molecules bind to the spike protein in a process called glycosylation, which plays an important role in the life cycle of the virus and has the ability to suppress the immune system. Their map included three predicted but never-before-seen glycosylation sites.
Although a German group has used a similar method to digitally extract images of the spike protein from SARS-CoV-2, Jin said, they had to first process the virus in formaldehyde to avoid there would be a risk of it taking anyone, and this treatment can cause chemical changes that prevent seeing the true structure.
Going forward, she said, the team would like to know how the part of the spike that binds to receptors on human cells is activated, and also use the same technique to study on spike proteins from the COVID-19-induced virus, which would require special biohazard resources.
COVID-19 vaccines target the spike protein – but this is another target
Provided by SLAC National Acceleration Laboratory
Citation: Scientists still get the most realistic view of coronavirus spike protein structure (2020, December 18) retrieved December 18, 2020 from https://phys.org/news/2020-12-scientists-realistic-view-coronavirus -spike.html
This document is subject to copyright. Other than any fair treatment for the purpose of scrutiny or private investigation, no part may be reproduced without written permission. The content is provided for informational purposes only.