
Embryos start out as single cells, and have to go from there to a complex series of multiple tissues. For organisms such as insects or frogs, this process is very easy to study, as development takes place in an egg that is deposited into the environment shortly after fertilization. But for mammals, where all development takes place within the reproductive field, understanding the earliest stages of development is a major challenge. It is difficult to perform any tests on a developing embryo, and effectively impossible at some stages.
This week, however, progress was made with both human and mouse embryos. On the human side, researchers have used stimulated stem cells to create embryo-like groups that perform the first major step in development, capturing them up to where mouse research has taken place. for decades. On the mouse side, however, a research team has forced mouse cells to fly for nearly a week outside the uterus. While this will open up a world of testing that has not been possible before, the requirements for taking on work mean that it is unlikely to be widely accepted.
To grow mice, you need rats
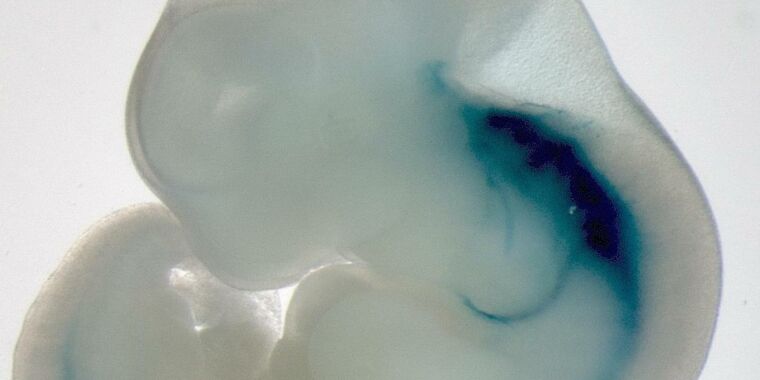
Mouse work was a little more technically interesting, so let’s get to that first. One of the most crucial steps in the development of the spine is called gastrulation. The process takes some cells that have been set aside early in the embryo, and turns them into three essential layers that go on to form the embryo: the skin and nerves, the lining of the embryo. cracking, and everything else.
Gastrulation occurs between six and seven days after fertilization, and many major developmental events occur shortly thereafter: the formation of zero cells and their organization, the development of the ordered structures that make up the vertebrae, and more. However, given the size of the embryo at this time and where it is located in the uterus, the process of gastrulation is largely invisible.
A large Israeli-based team decided to figure out how that would change. They first started with embryos that had already passed the required levels of gastrulation, and worked out how to survive. It was … it’s not simple. Initially, embryos had to be packed in a constantly rotating bottle to ensure that all the nutrients and oxygen around the embryo were mixed. in detail, instead of allowing the energy demands of the embryo to create a local “dead zone” around it.
Oxygen levels had to be controlled through a standard internal gas supply system that stimulated the pressure over time to bring more oxygen into solution. And fresh glucose had to be regularly introduced into the melting medium as well.
About that melting medium. About a quarter of it was something you could buy out of a typical biotechnology supply catalog. The rest were much harder to buy. Half of it was given from rat blood. And a quarter of serum was obtained from human umbilical cord blood. None of these are particularly easy to come by. They tested, and you need human blood; rat blood alone was not nearly as good. (How often can I write a sentence like that?)
In any case, this was enough to get the embryos through four days of development. This led them from three layers of nonspecific cells to a place where the spine had begun to form and limbs began to come off the side of the embryo. That, in terms of development, involves a lot of important events that we are very interested in exploring.
However, by this point, the circulating system of growing embryos should have been integrated with the placenta, ensuring that the entire embryo was full of nutrients and oxygen. The embryos died in a way that showed a lack of oxygen supply.
Earlier still …
While in performance, all this happens after gastrulation occurs. Thus, the researchers reintroduced support a little longer, separating embryos between four and five days after fertilization. The same medium worked, but here, the embryos did not need to be in a rotating bottle to survive. The two stimuli could be combined, essentially carrying the embryos through a week of complete development outside the uterus.
In addition, the team showed that they were able to differentiate the embryos during this period in culture. This involved inserting DNA into their cells (using either virus or electrical currents), or inserting stem cells to see how they develop. So, for anyone willing to get enough rats and umbilical blood for this to work, there is a lot of developmental biology study that can now be done on mouse embryos.
The only thing that is not accessible through this work is the earliest developmental process, in which a cavity opens up in the organ of uniform-like cells formed by the first cell sections of the fertilized egg. . This creates the first rather distinct numbers of cells in the embryo (both the outer ones, and a bunch of cells inside the ovary). The resulting structure is called a blastocyst.
For mice, we are able to take a fertilized egg and grow it into an explosion in culture for years. But this was not done by human cells. And to some extent, not yet. Instead, two different scales started with stem cells, either embryonic stem cells or stem cells taken from adult figs. In contrast to mouse activity, the embryo could go so far as to do this with off-the-shelf ingredients in the media in which the cells were grown.
This opens the earliest stages of human development for study. However, while the formation of the blastocyst is interesting, much more will take place at later stages of development. And here, ethical concerns tend to limit the extent to which we are willing to endure human tension in culture.
The interesting thing here is that we can already get mouse embryos to become blastocysts, and now we can take blastocysts along the way through development. So it looks like we might be able to combine the two processes with some work. That would be enough to get from fertilization to about two-thirds of the way to birth. That is very impressive.
But by this point, the embryo becomes very three-dimensional, and getting all the cells supplied with oxygen and nutrients really requires a functioning blood supply. , plugged into a store for the needs of the embryos. And it’s not clear how we could replace the placenta, which maintains a very complex and specialized exchange between the bloodstream of the fetal and maternal. In practice, however, these results mean that it is now possible to test a full range of experiments in mice – as long as you are willing to bleach enough rats.
Nàdar, 2021. DOI: 10.1038 / s41586-021-03416-3, 10.1038 / s41586-021-03372-y, 10.1038 / s41586-021-03356-y (Mu DOIs).